Oxymercuration reduction Study guides, Class notes & Summaries
Looking for the best study guides, study notes and summaries about Oxymercuration reduction? On this page you'll find 12 study documents about Oxymercuration reduction.
All 12 results
Sort by

-
Test Bank for Organic Chemistry, 4th Edition by David R. Klein | 9781119659594|Chapter 1-27 | All Chapters with Answers and Rationals
- Exam (elaborations) • 2151 pages • 2023
- Available in package deal
-
- $24.14
- 16x sold
- + learn more
Test Bank for Organic Chemistry 4e 4th Edition by David R. Klein. ISBN-13: 9594 Full chapters test bank PDF TABLE OF CONTENTS 1 A Review of General Chemistry: Electrons, Bonds, and Molecular Properties 1 1.1 Introduction to Organic Chemistry 2 1.2 The Structural Theory of Matter 3 1.3 Electrons, Bonds, and Lewis Structures 4 1.4 Identifying Formal Charges 7 1.5 Induction and Polar Covalent Bonds 8 1.6 Reading Bond-Line Structures 11 1.7 Atomic Orbitals 14 1.8 Valence Bond Theory 17 1.9 Molecular...

-
chem 210 exam 3 with all questions correctly answered
- Exam (elaborations) • 5 pages • 2023
-
Available in package deal
-
- $13.49
- + learn more
less the more s character a carbocation has, the _____stable it is the same as when looking at an epoxide, the orientation around the oxygen is _________ the orientation around the double bond in the reactant BrOH when added with base makes an epoxide Br2 adds 2 Brs in anti conformation lindlar catalyst produces a Z alkene from an alkyne (adds 2 Hs in syn) add more electronegative atom, add pi bond 2 ways of producing an oxidation reaction remove e...

-
Test Bank for Organic Chemistry, 4th Edition by David R. Klein
- Exam (elaborations) • 2149 pages • 2022
-
- $49.99
- 75x sold
- + learn more
Test Bank for Organic Chemistry 4e 4th Edition by David R. Klein. ISBN-13: 9594 Full chapters test bank PDF TABLE OF CONTENTS 1 A Review of General Chemistry: Electrons, Bonds, and Molecular Properties 1 1.1 Introduction to Organic Chemistry 2 1.2 The Structural Theory of Matter 3 1.3 Electrons, Bonds, and Lewis Structures 4 1.4 Identifying Formal Charges 7 1.5 Induction and Polar Covalent Bonds 8 1.6 Reading Bond-Line Structures 11 1.7 Atomic Orbitals 14 1.8 Vale...

-
Chem 210 Exam 3 Questions with Complete Solutions Rated A+
- Exam (elaborations) • 4 pages • 2023
-
Available in package deal
-
- $11.99
- + learn more
less - the more s character a carbocation has, the _____stable it is the same as - when looking at an epoxide, the orientation around the oxygen is _________ the orientation around the double bond in the reactant BrOH - when added with base makes an epoxide Br2 - adds 2 Brs in anti conformation lindlar catalyst - produces a Z alkene from an alkyne (adds 2 Hs in syn) add more electronegative atom, add pi bond - 2 ways of producing an oxidation reaction remove e...

-
Test Bank for Organic Chemistry, 4th Edition by David R. Klein 9781119659594 | All Chapters with Answers and Rationals
- Exam (elaborations) • 2151 pages • 2023
- Available in package deal
-
- $22.81
- + learn more
Test Bank for Organic Chemistry 4e 4th Edition by David R. Klein. ISBN-13: 9594 Full chapters test bank PDF TABLE OF CONTENTS 1 A Review of General Chemistry: Electrons, Bonds, and Molecular Properties 1 1.1 Introduction to Organic Chemistry 2 1.2 The Structural Theory of Matter 3 1.3 Electrons, Bonds, and Lewis Structures 4 1.4 Identifying Formal Charges 7 1.5 Induction and Polar Covalent Bonds 8 1.6 Reading Bond-Line Structures 11 1.7 Atomic Orbitals 14 1.8 Valence Bond Theory 17 1.9 Molecular...

-
Unit 3 :CHEM 210 with 100% correct answers
- Exam (elaborations) • 4 pages • 2023
-
Available in package deal
-
- $13.99
- + learn more
Bronsted-Lowry Acid Must Have a Proton (H) Bronsted-Lowry Base Must have Lone Pairs or Pi Bonds Neutralization Strong Acid donates a proton to the Strong Base to generate Water and the Conjugate Base Electrophile Electron Loving An atom that wants electrons. An atom that is electron rich. Must have a partial positive charge. (Cation) Nucleophile Nucleus Loving If opposites attract, then a _________ is electron rich. Must have a partial negative charge. (An...

-
Test Bank for Organic Chemistry 4th Edition by David R. Klein | 9781119659594 |2021-2022 |Chapter 1-27 | All Chapters with Answers and Rationals
- Exam (elaborations) • 2151 pages • 2023
-
- $42.98
- + learn more
Test Bank for Organic Chemistry 4e 4th Edition by David R. Klein. ISBN-13: 9594 Full chapters test bank PDF TABLE OF CONTENTS 1 A Review of General Chemistry: Electrons, Bonds, and Molecular Properties 1 1.1 Introduction to Organic Chemistry 2 1.2 The Structural Theory of Matter 3 1.3 Electrons, Bonds, and Lewis Structures 4 1.4 Identifying Formal Charges 7 1.5 Induction and Polar Covalent Bonds 8 1.6 Reading Bond-Line Structures 11 1.7 Atomic Orbitals 14 1.8 Valence Bond Theory 17 1.9 Molecular...

-
DAT Organic Chemistry Reactions
- Exam (elaborations) • 13 pages • 2023
-
- $18.99
- + learn more
--NO2 + Sn/HCl - Correct Answers>Clemmenson Reduction --NH2 1,2-addition conj double bonds + RMgX or LiAlH4 - Correct Answers>Nucleophiles attack the carbonyl directly. Strong nucleophiles: Gringards, NaBH4, LiAlH4 1. (2) esters CH3--C=O--OEt + NaOR, EtOH 2. H3O+ ketone + (ester) CH3--C=O--OEt + -OR + H3O+ --> - Correct Answers>Claisen Condensation enolate ion of one ester acts as nucleophile attacking another ester 1. NaOR, EtOH 2. H3O+ Anhydride for mixed 1. BH3/TH...
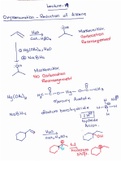
-
Oxymercuration-Reduction of Alkene Notes
- Class notes • 4 pages • 2023
- Available in package deal
-
- $4.49
- + learn more
Notes over oxymercuration-reduction of alkene and its mechcanism.

-
Blank Oxymercuration-Reduction Mechcanism Sheet
- Class notes • 1 pages • 2023
- Available in package deal
-
- $2.99
- + learn more
Blank mechanism sheet for oxymercuration-reduction

Did you know that on average a seller on Stuvia earns $82 per month selling study resources? Hmm, hint, hint. Discover all about earning on Stuvia


